ABOUT
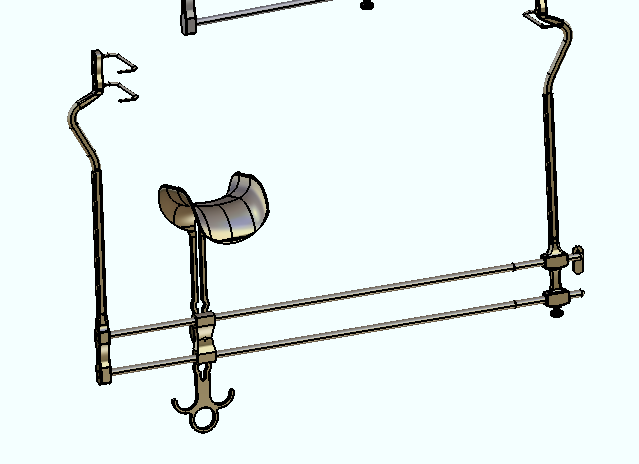
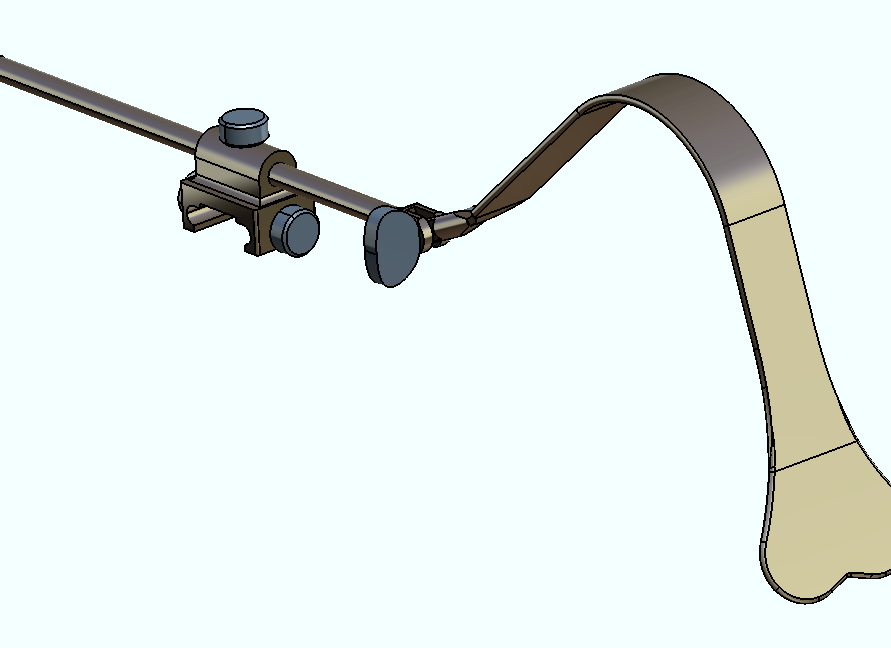
For summer 2017, I interned at Aesculap Implant Systems in the R&D Customized Instruments department (Breinigsville, PA). Aesculap is a German company whose U.S. base focuses on spinal, orthopedic, and neural surgical instrumentation markets.
Contributions
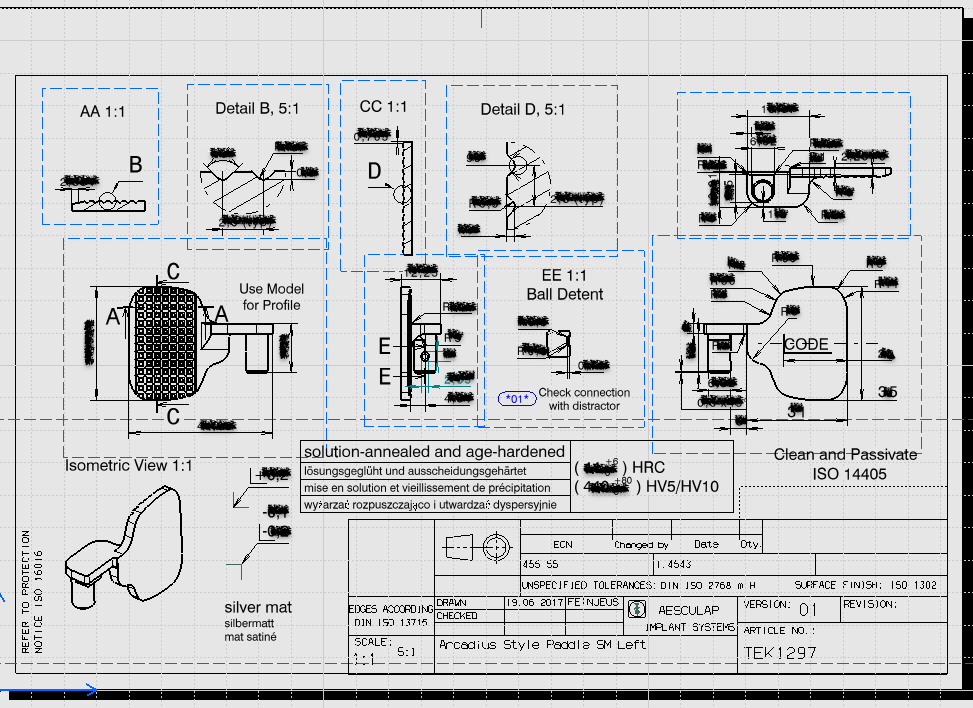
My work involved modifying existing surgical instruments or prototyping designs from abstract concept from surgeon requests. I created engineering models and drawings in CATIA V5 from scratch, using just a ruler, optical comparator, and calipers! From those models, I generated proper engineering drawings using GD&T for our in-house machinists. They would walk me through different machining options for the ~30 projects I worked on, and I gained plenty of insight from the accompanied cost-benefit analysis for time, cost, and material choice for manufacturing.
On the documentation side, I completed design history files for the FDA 510(k) process (per 21 CFR 820) to ensure proper biocompatibility, analyze possible failure modes (FMEA), and confirm functionality.
SKILLS
CATIA V5 modeling, Design for manufacturing and assembly, GD&T, FMEA, FDA documentation (21 CFR 820), Spinal and orthopedic surgical procedures, implants, and equipment